KK Bldg. 6F 1-4-11 Higashi Kanda, Chiyoda-ku,
Tokyo,101-0031 Japan
TEL +81-3-3864-5570, FAX +81-3-5821-7559
- English | - Japanese
Tokyo,101-0031 Japan
TEL +81-3-3864-5570, FAX +81-3-5821-7559
- English | - Japanese
1. What is activated carbon?

Powdered Carbon

Granular Carbon

Pelleted Carbon
A mixture of crystalline and amorphous structures within the carbon, resembling a hard sponge.
This adsorbent is not a chemically synthesized product. It is environmentally friendly and can be reused after regeneration. Pores are distributed within the carbon. Physical adsorption occurs through van der Waals forces.
This adsorbent is not a chemically synthesized product. It is environmentally friendly and can be reused after regeneration. Pores are distributed within the carbon. Physical adsorption occurs through van der Waals forces.
2. Adsorption
The amount of adsorption varies depending on the impurity concentration. Impurities are adsorbed to the surface of activated carbon through physical adsorption, generally adsorbing organic matter but not inorganic matter.
The amount of organic matter adsorbed varies depending on the target substance, but is generally 10-30 wt%. This means that 100 g of activated carbon can remove 10-30 g of impurities.
Naturally, the amount of adsorption varies greatly depending on the conditions, so it is important to process under optimal adsorption conditions.
The amount of organic matter adsorbed varies depending on the target substance, but is generally 10-30 wt%. This means that 100 g of activated carbon can remove 10-30 g of impurities.
Naturally, the amount of adsorption varies greatly depending on the conditions, so it is important to process under optimal adsorption conditions.
Illustration of Adsorption
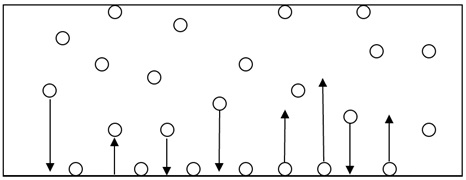
Impurities are adsorbed onto the surface of activated carbon through intermolecular attraction. The equilibrium state is reached when the amount of impurity molecules attracted to the surface and the amount detached from it are equal.
Therefore, as the amount of impurities in the liquid increases, the adsorption capacity also increases.
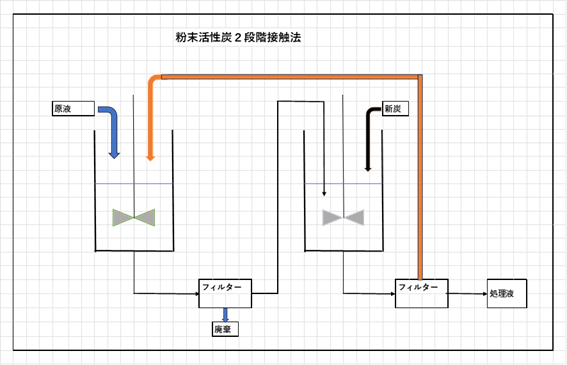
With powdered activated carbon, adsorption terminates when equilibrium is reached after adding activated carbon in batch treatment, meaning only about 60% of the adsorption capacity can be effectively utilized.
To maximize adsorption, methods such as two-stage contact (adding used powdered activated carbon back to the treated liquid) are used. See the diagram below.
Therefore, as the amount of impurities in the liquid increases, the adsorption capacity also increases.
With powdered activated carbon, adsorption terminates when equilibrium is reached after adding activated carbon in batch treatment, meaning only about 60% of the adsorption capacity can be effectively utilized.
To maximize adsorption, methods such as two-stage contact (adding used powdered activated carbon back to the treated liquid) are used. See the diagram below.
In contrast, granular carbon always exhibits a higher concentration at the inlet, allowing it to achieve the maximum adsorption capacity for a given liquid.
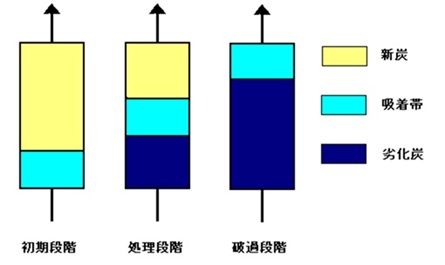
Adsorption begins at the inlet and gradually moves toward the outlet. The adsorption zone is called the adsorption zone (Mass Transfer Zone, or MTZ).
The point at which impurities begin to leak from the adsorption tower is called breakthrough. At breakthrough, a portion of the adsorption zone still retains the adsorption capacity of the activated carbon, rendering that portion unusable.
Therefore, the tower's design conditions require a safety factor of 70% or 80% to calculate the throughput. See the diagram below for the movement of the adsorption zone.
Adsorption begins at the inlet and gradually moves toward the outlet. The adsorption zone is called the adsorption zone (Mass Transfer Zone, or MTZ).
The point at which impurities begin to leak from the adsorption tower is called breakthrough. At breakthrough, a portion of the adsorption zone still retains the adsorption capacity of the activated carbon, rendering that portion unusable.
Therefore, the tower's design conditions require a safety factor of 70% or 80% to calculate the throughput. See the diagram below for the movement of the adsorption zone.
3. Pores
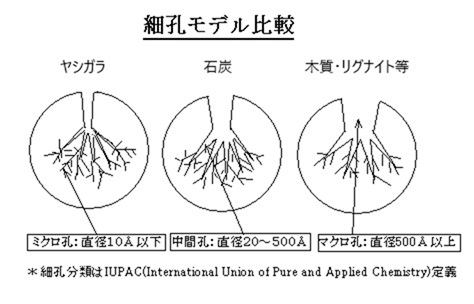
Adsorption occurs primarily in activated carbon's micropores. In the liquid phase, adsorption also occurs in mesopores.
Macropores are involved in the adsorption rate (adsorption speed). The adsorption capacity of activated carbon is tested using iodine, methylene blue, caramel, etc., but each test measures areas with different pore sizes.
Iodine adsorption is associated with small micropores, methylene blue with large micropores, and caramel with larger mesopores.
Differences in pore size distribution can have a significant impact depending on the size of the impurities to be removed. For example, when decolorizing sugar, the color molecules are too large to enter the micropores, so coconut shell activated carbon, which has a highly developed micropore system, cannot decolorize sugar, no matter how large its surface area (e.g., 2000 m2/g).
Pore size distribution is determined by the raw materials and manufacturing method, so the raw materials are extremely important.
Macropores are involved in the adsorption rate (adsorption speed). The adsorption capacity of activated carbon is tested using iodine, methylene blue, caramel, etc., but each test measures areas with different pore sizes.
Iodine adsorption is associated with small micropores, methylene blue with large micropores, and caramel with larger mesopores.
Differences in pore size distribution can have a significant impact depending on the size of the impurities to be removed. For example, when decolorizing sugar, the color molecules are too large to enter the micropores, so coconut shell activated carbon, which has a highly developed micropore system, cannot decolorize sugar, no matter how large its surface area (e.g., 2000 m2/g).
Pore size distribution is determined by the raw materials and manufacturing method, so the raw materials are extremely important.
| 4. | Differences Due to Shape |
| (Powder/Granular) |
Since activated carbon adsorption occurs through internal pores, the external shape does not affect the adsorption capacity.
However, differences do occur depending on actual usage conditions. For example, if the treatment time (contact time) of activated carbon is short, powdered carbon will have a higher adsorption capacity. This is because powdered charcoal, with its fine particles, has high contact efficiency, reaching equilibrium in 30 to 40 minutes even in the liquid phase, whereas granular charcoal requires several hours of contact. There is no difference in the amount of adsorption at equilibrium between the two.
The general differences are as follows:
However, differences do occur depending on actual usage conditions. For example, if the treatment time (contact time) of activated carbon is short, powdered carbon will have a higher adsorption capacity. This is because powdered charcoal, with its fine particles, has high contact efficiency, reaching equilibrium in 30 to 40 minutes even in the liquid phase, whereas granular charcoal requires several hours of contact. There is no difference in the amount of adsorption at equilibrium between the two.
The general differences are as follows:
| Powdered: | Batch process, short processing time, low equipment costs, flexibility to accommodate fluctuations in feedstock solution, poor energy efficiency, non-recyclable |
| Granular: | Continuous process, long processing time, high equipment costs, inflexibility to accommodate fluctuations in feedstock solution, good energy efficiency, recyclable |
| * | Because granular charcoal can be regenerated, it is often worth the investment in equipment when consumption is high. When switching from powdered charcoal to granular charcoal, granular charcoal is considered preferable if consumption is 20 tons or more per month. |
| HOME
| Product Information
- Granular activated carbon (coconut shell raw material)
- Granular activated carbon (coal raw material)
- Powdered activated carbon
- Special charcoal (impregnated charcoal)
| Technical information | Company information | Contact | Handling of personal information
| Product Information
- Granular activated carbon (coconut shell raw material)
- Granular activated carbon (coal raw material)
- Powdered activated carbon
- Special charcoal (impregnated charcoal)
| Technical information | Company information | Contact | Handling of personal information
 Obbro Japan Co.,Ltd.
Obbro Japan Co.,Ltd.